Chemical Reactivity
|
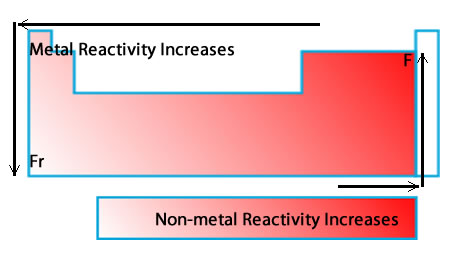
Metals:
· Reactivity increases as you go down a group because the farther down you go, the easier it is for electrons to be given/taken · Reactivity decreases as you go across a period because there is more electrons to get rid of, which need more energy Non-Metals: · Reactivity increases as you go up a group because the higher up and to the right the elements are, the more chances of an exchange of electrons · Reactivity increases as you go across a period because the close the elements get to filling their orbitals the more motivated they are to do so |