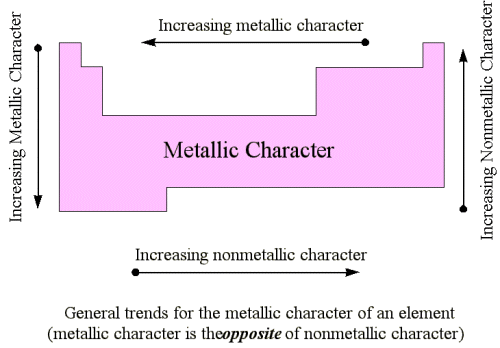
Metallic Character based on its position on the Periodic table
|
The metallic character of an element can be determined by its position in the periodic table. As you move towards the left of the periodic table, the metallic character of the elements increases. Elements near the bottom of the periodic table have higher metallic character than those at the top. This is why metalloids closer to the left and the bottom of the periodic table share more properties with metals and those nearer to the right and top with non-metals.
|